Additionally, aflibercept biosimilars under development include FYB203 (Formycon AG, Munich, Germany and Bioeq Gmbh Holzkirchen, Germany), ALT-L9 (Alteogen, Deajeon, South Korea), MYL1710 (Momenta Pharamaceuticals, Cambridge, MA, and Mylan Pharmacueticals, Canonsburg, PA), CHS-2020 (Coherus BioSciences, Redwood City, California). 
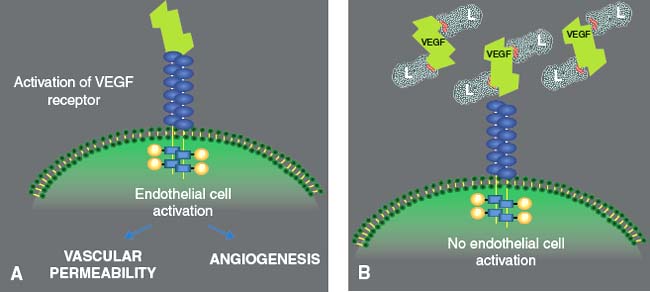
Cheaper alternatives, including ranibizumab biosimilars, include razumab (Intas Pharmaceuticals Ltd., Ahmedabad, India), FYB 201 (Formycon AG, Munich, Germany and Bioeq Gmbh Holzkirchen, Germany), SB-11 (Samsung Bioepsis, Incheon, South Korea), xlucane (Xbrane Biopharma, Solna, Sweden), PF582 (Pfnex, San Diego, California), CHS3551 (Coherus BioSciences, Redwood City, California). Anti-VEGF agents have revolutionized our treatment of wet AMD however, real world studies have shown limited visual improvement in patients over time, largely due to the large treatment burden. Currently, the major therapies in AMD focus on the VEGF-A pathway, of which the most common are bevacizumab (Avastin Genentech, San Francisco, California), ranibizumab (Lucentis Genentech, South San Francisco, California), and aflibercept (Eylea Regeneron, Tarrytown, New York). Prior treatments have included focal laser therapy, verteporfin (Visudyne, Bausch and Lomb, Rochester, New York) ocular photodynamic therapy, transpupillary thermotherapy, intravitreal steroids and surgical excision of choroidal neovascular membranes. There are various cytokine pathways involved in the formation and leakage from CNV. Neovascular AMD comprises 10% of all cases and can lead to devastating visual loss due to choroidal neovascularization (CNV). Age related macular degeneration (AMD) is the most common cause of vision loss in the elderly population.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
